The primary objective of dispersing pigments is to separate the pigment agglomerates that are formed during the drying processes. This is done in a dispersion or grinding medium by mechanical means.
As particle size is reduced, the surface area increases, leading to an improvement in optical properties, such as tinctorial strength, gloss, brightness, opacity or transparency.
01
Why are Dispersants Important?

02
The Role of Hyperdispersants
Solsperse™ Hyperdispersants have two fundamental roles in surface coatings: improving pigment dispersion and reducing interparticulate attraction. This leads to a smaller average particle size with a narrower particle size distribution.
Small particles are generally more prone to reagglomeration or flocculation. With dispersants reducing interparticle attraction, however, dispersions are significantly more stable.




03
Background to Effective Stabilization
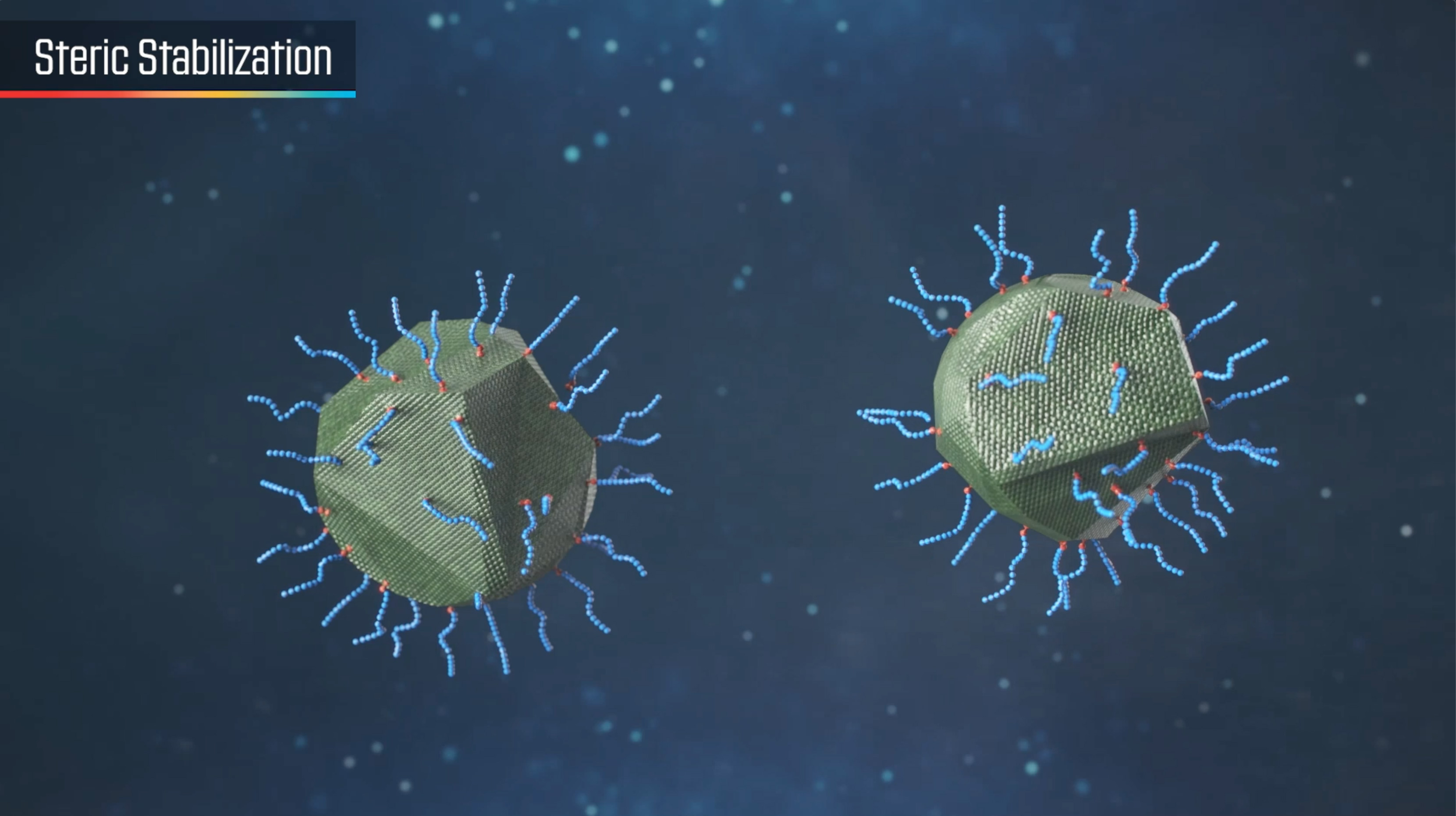
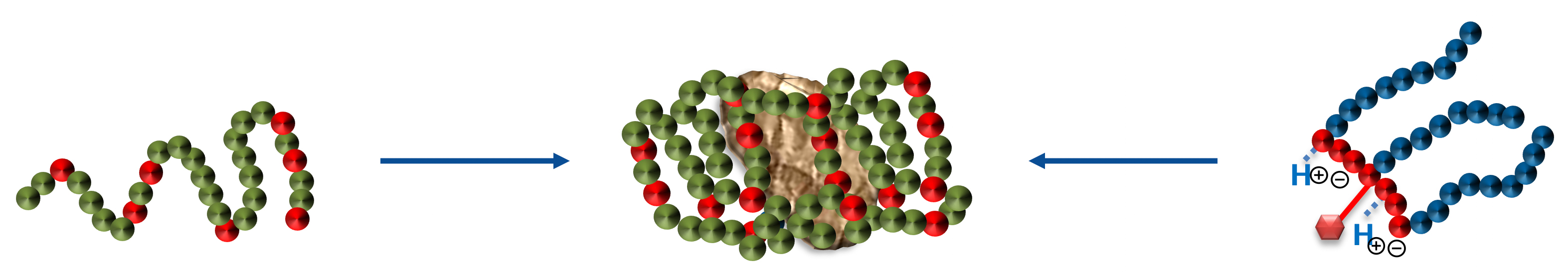
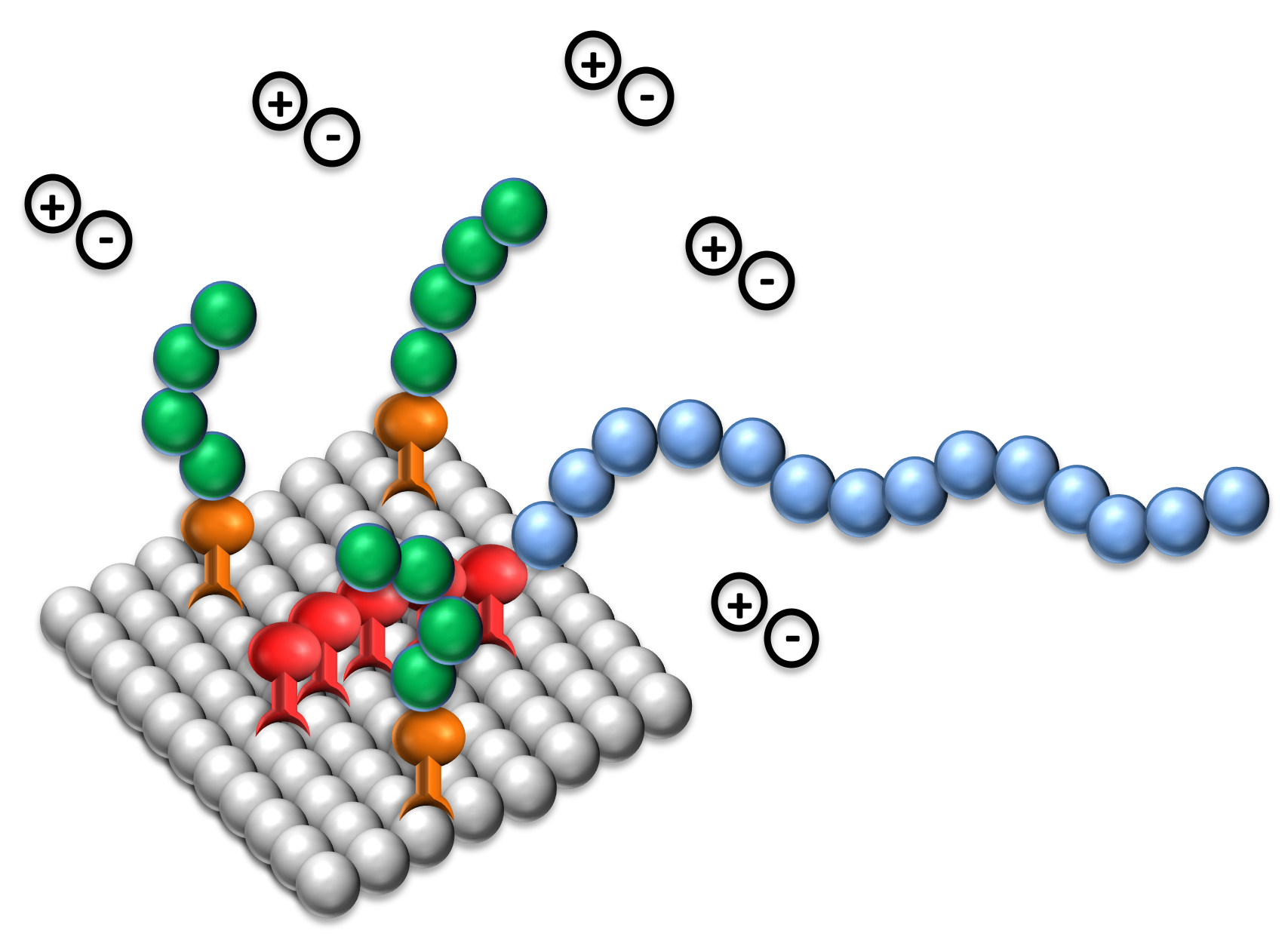
There are two principal mechanisms for the stabilization of pigmented dispersions: electrostatic stabilization and steric stabilization. Both require the adsorption of stabilizing molecules on the pigmented surface.
STERIC STABILIZATION
04
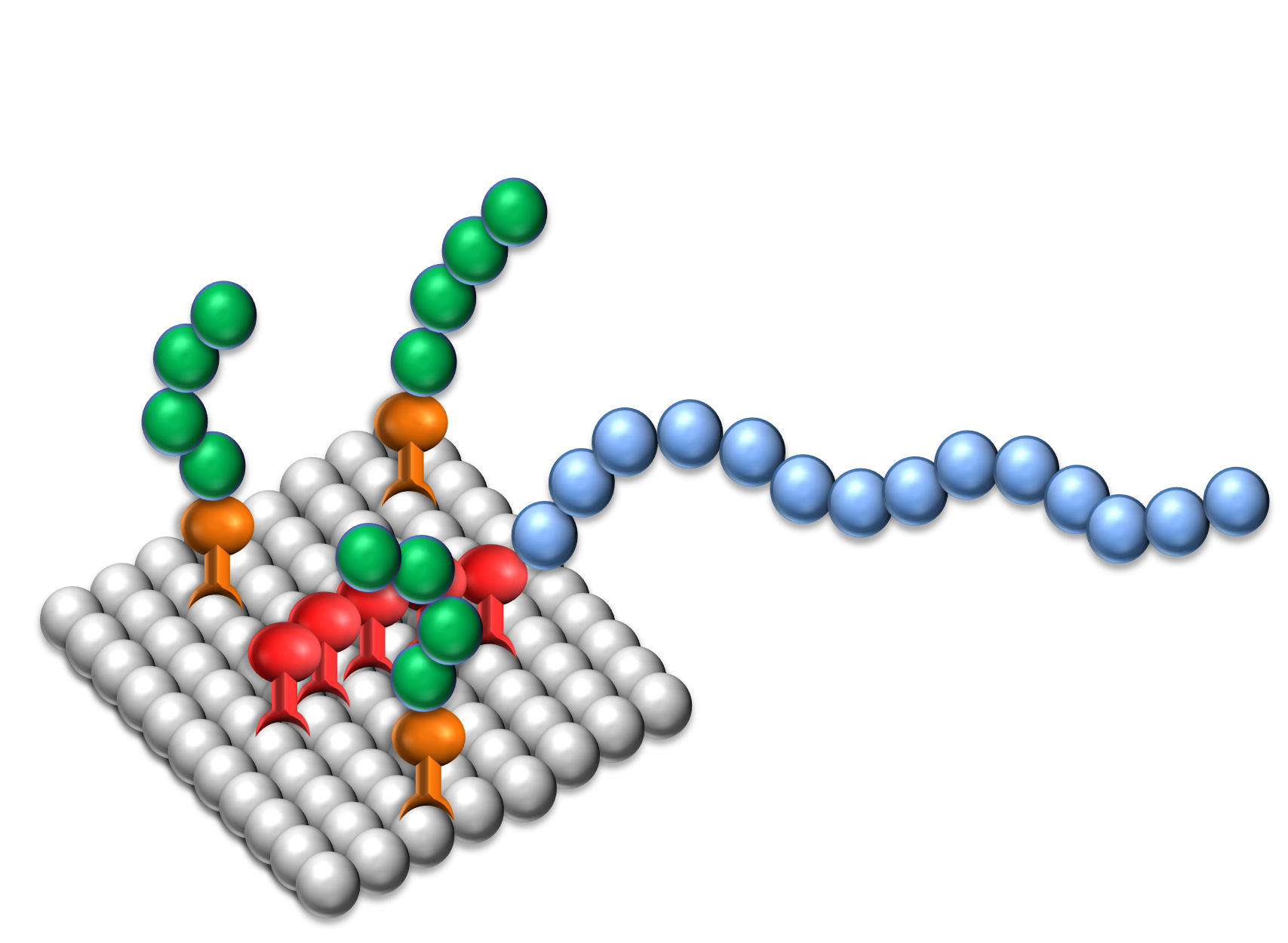
The Design of Dispersants
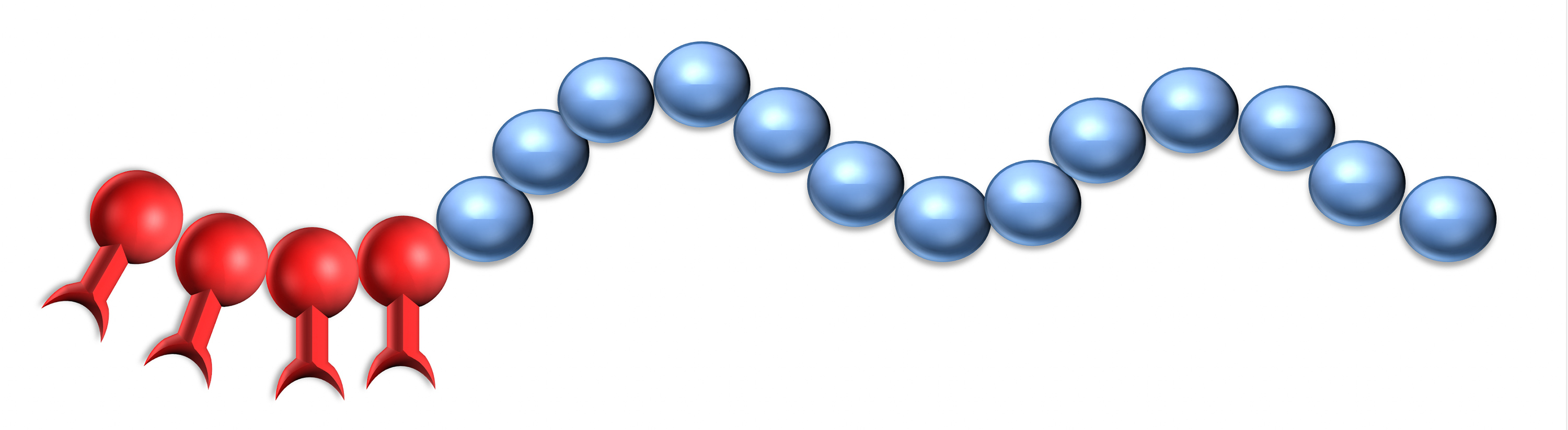
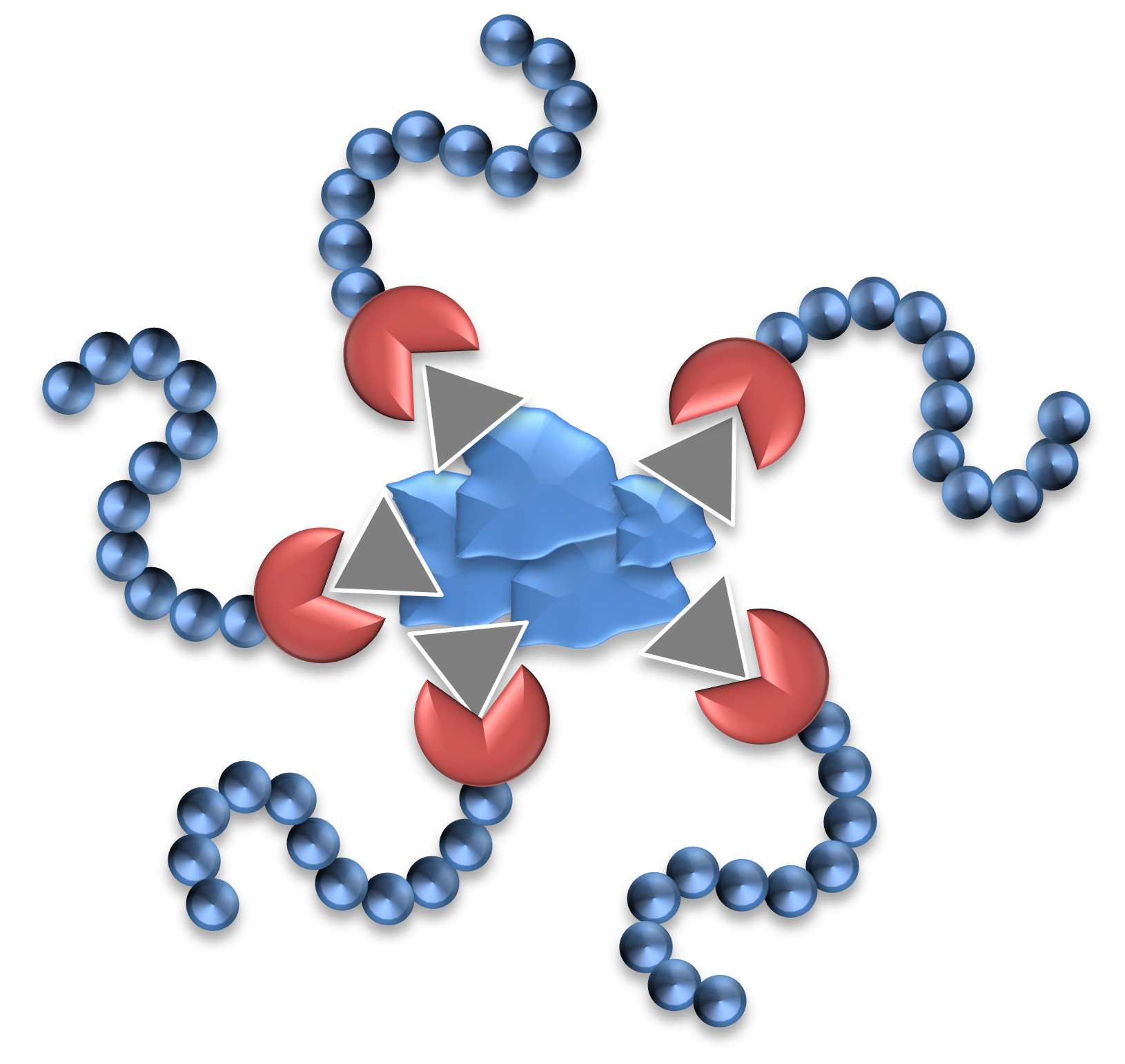
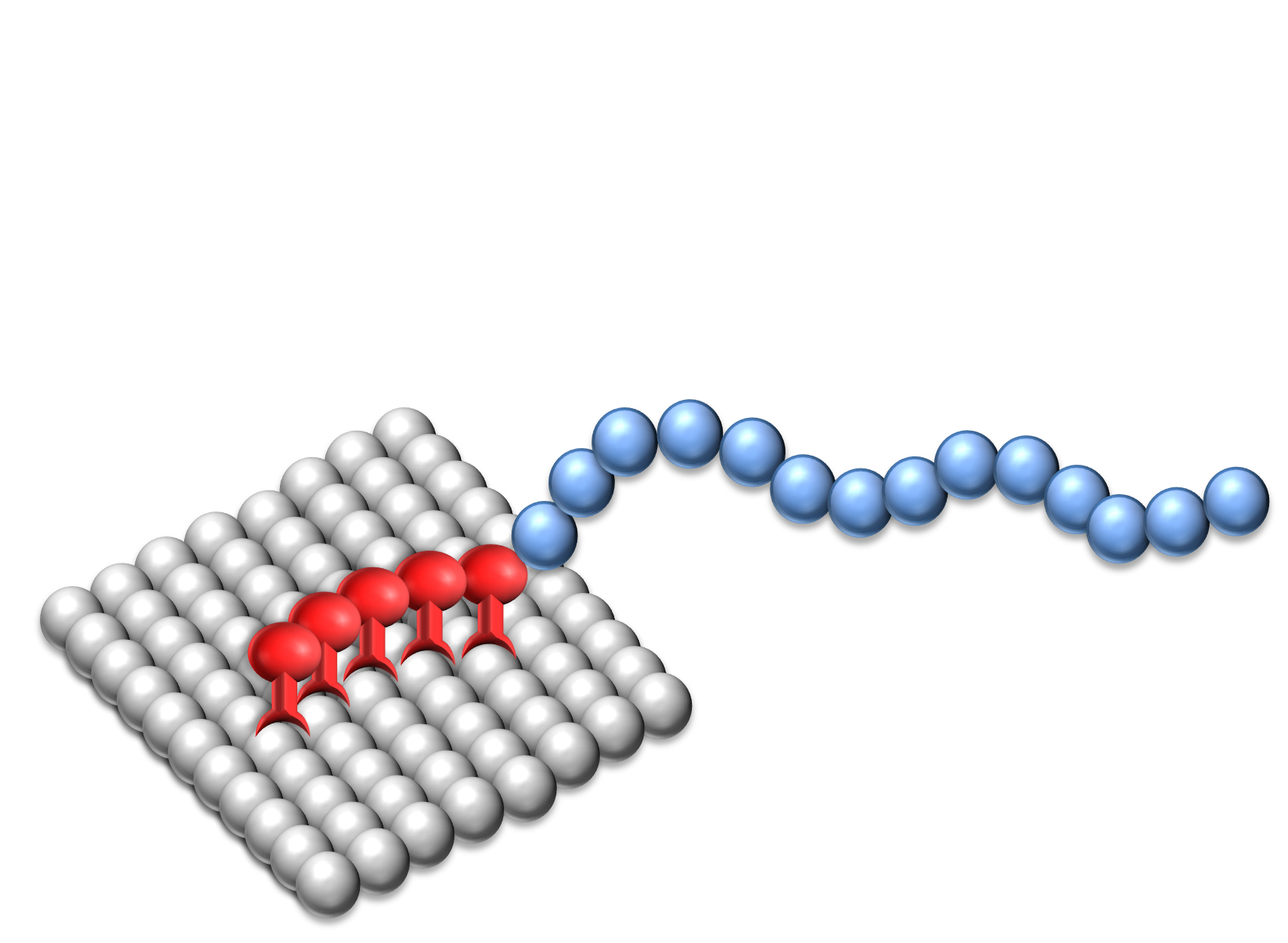
Dispersants are two-component structures: the anchoring group providing strong adsorption onto the pigment surface and polymeric chains attached to the group delivering stabilization.
The effectiveness of Solsperse™ Hyperdispersants is caused by the particular combination of anchoring group and polymeric chains.
05
Anchor Groups
It does not matter whether the polymer chains are provided by dispersants containing single chains or up to many hundreds of chains; both types are available. What is essential is that the chains are successfully anchored to the pigment surface, and that the surface of the particles are covered with a sufficient density of chains to ensure minimum particle-to-particle interaction.
As the nature of a pigment‘s surface depends on its chemical type, many different chemical groups are used as anchor groups for dispersants. The Solsperse™ Hyperdispersants range contains several different types of anchor groups, varying from products only suitable for inorganic pigments, to those suitable for the whole range of pigments in use today. In dispersants for aqueous systems the anchor group is hydrophobic relative to the rest of the dispersant and also contains specific functionalities to interact with the pigment.
SYNERGISTS
With particularly difficult non-polar pigments, a very successful anchoring approach is the use of Solsperse™ synergists. The synergists are pigment derivatives with a strong affinity for certain pigment types that furnish the pigment surface with anchoring sites for the conventional polymeric Solsperse™ Hyperdispersants.
This versatile approach has enabled Lubrizol to produce a range of products with anchoring mechanisms that do not adversely interfere with the cure of reactive systems such as acid catalyzed, isocyanate cured or reactive monomer coatings. This is particularly important due to the growing importance of dispersants in tinters for mixing schemes, where many different resins are in use.
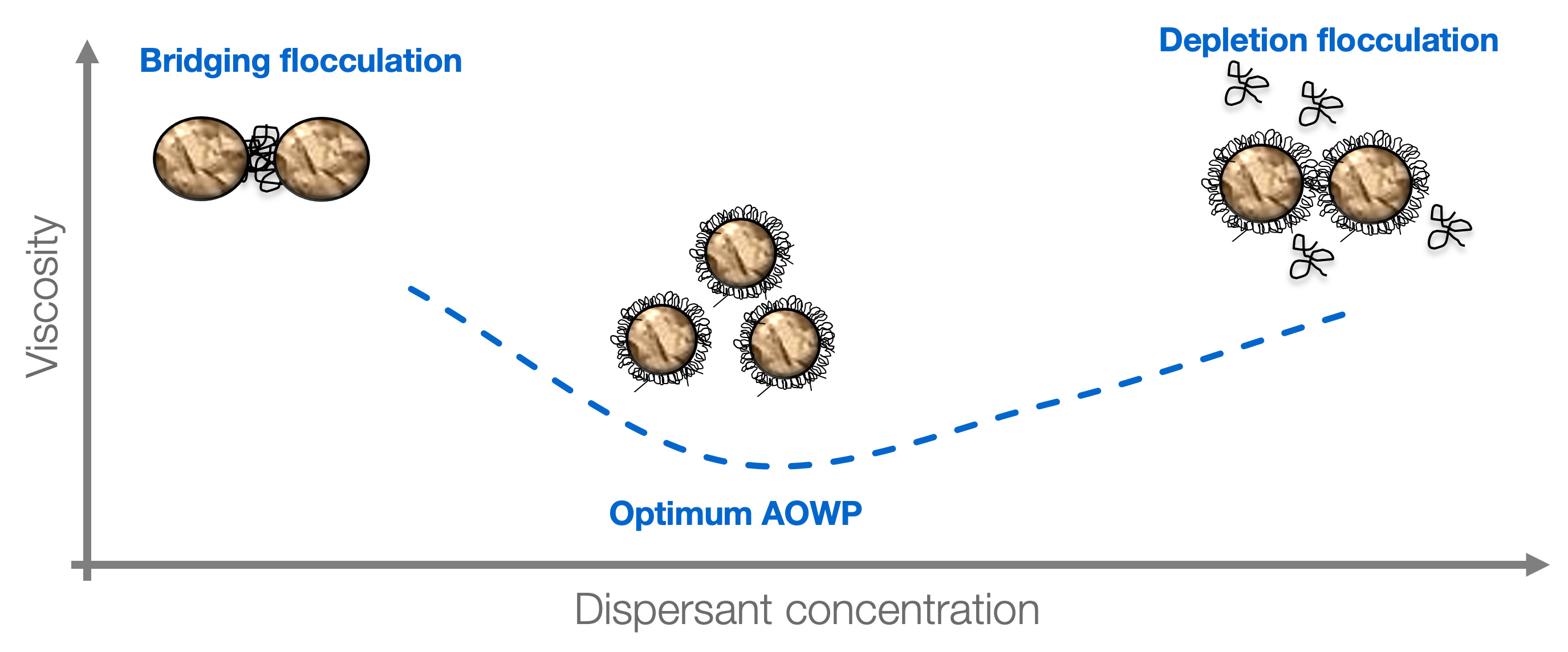
The optimum amount of Solsperse™ Hyperdispersant required is dependent on the surface area of the pigment. If too little is used, the full benefits will not be realized. If too much is used, the thickness of the protective barrier could be reduced as a result of overcrowding on the pigment surface.
An excess of dispersant leads to inferior final coating properties and film properties such as adhesion or hardness can be adversely affected because of the presence of free dispersant molecules in the drying film.
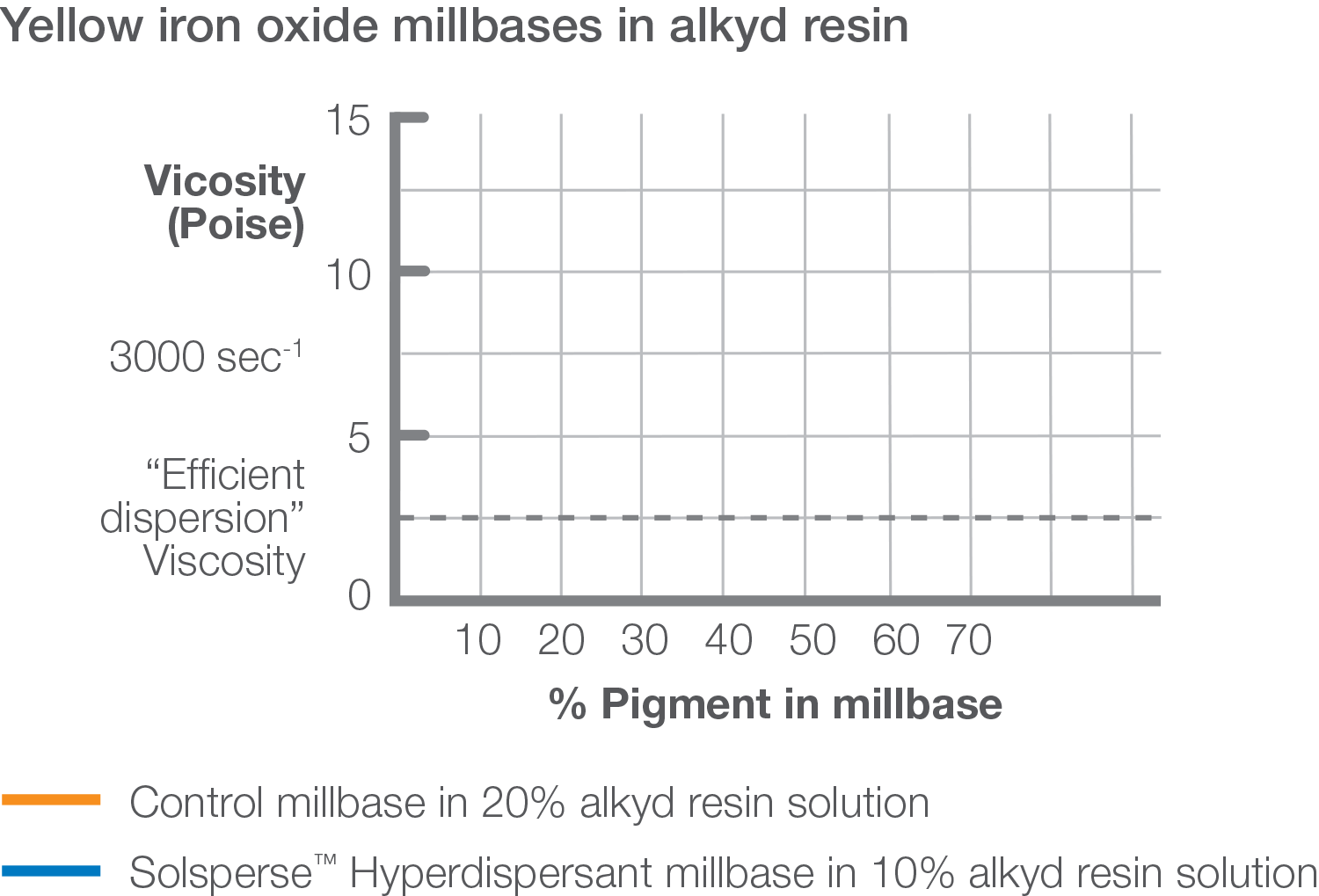
As a general rule 2mg of polymeric dispersant per square meter of pigment surface area will be close to the optimum amount required. A very simple calculation may be used to determine the amount of dispersant required, expressed as percentage Agent On Weight of Pigment (%AOWP).
A ladder series of polymeric dispersant levels should be carried out around this 2mg/m² level. Measurement of millbase viscosity will show a minimum at the optimum dosage. Alternatively, it’s possible to measure gloss or color strength of the coating, which will show a maximum at the same optimum dosage.
06
Practical Demonstration of Need for Correct Dosage
The optimum amount of Solsperse™ Hyperdispersant will not provide a ‘knife-edge’ change in any optical property but further additions are unlikely to result in extra improvements.
07
Formulation Guidelines When Using Solsperse™ Hyperdispersants
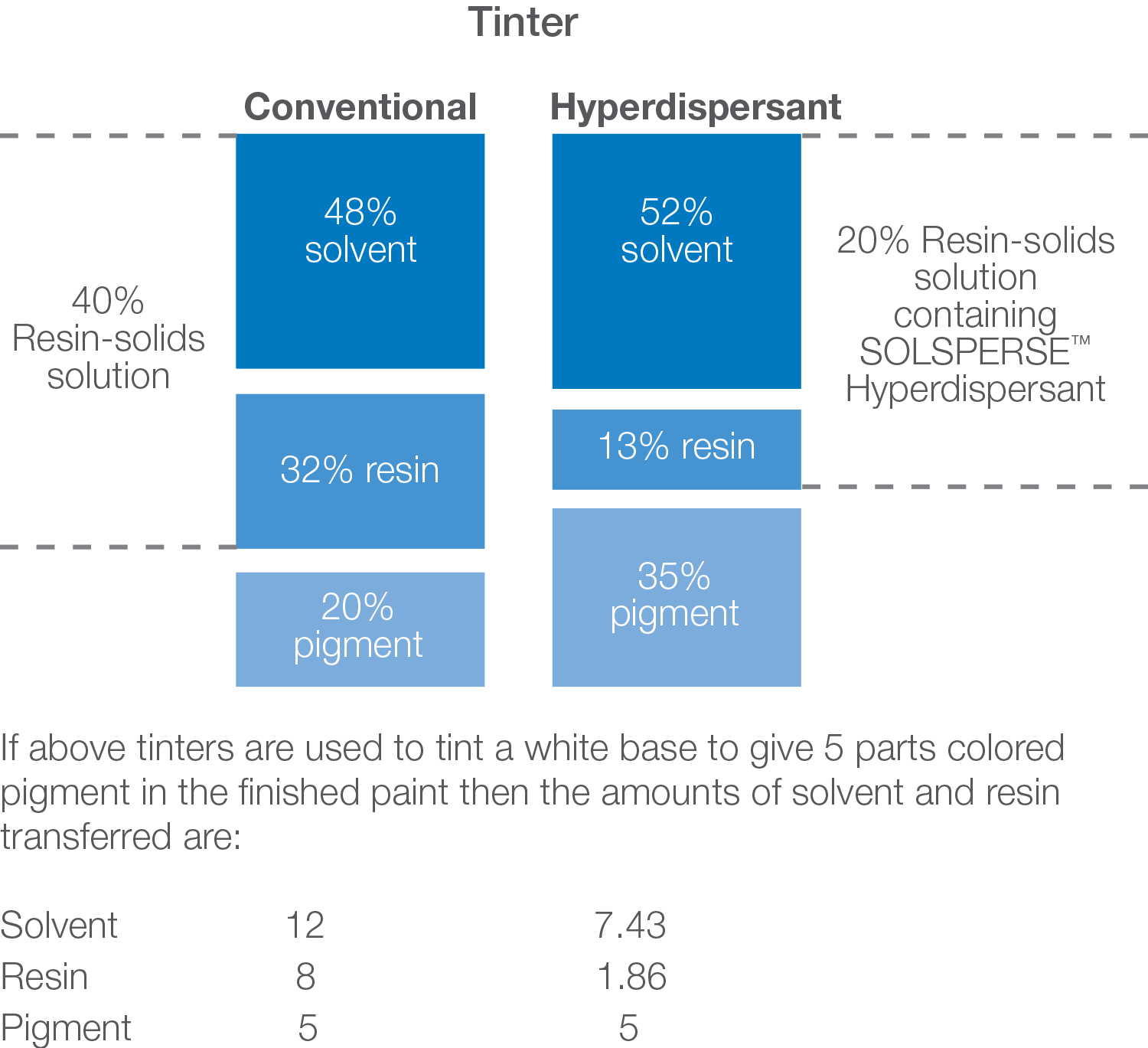
MINIMAL RESIN
Dispersants are widely used in applications where resins are a component of the millbase e.g. inks and paints.
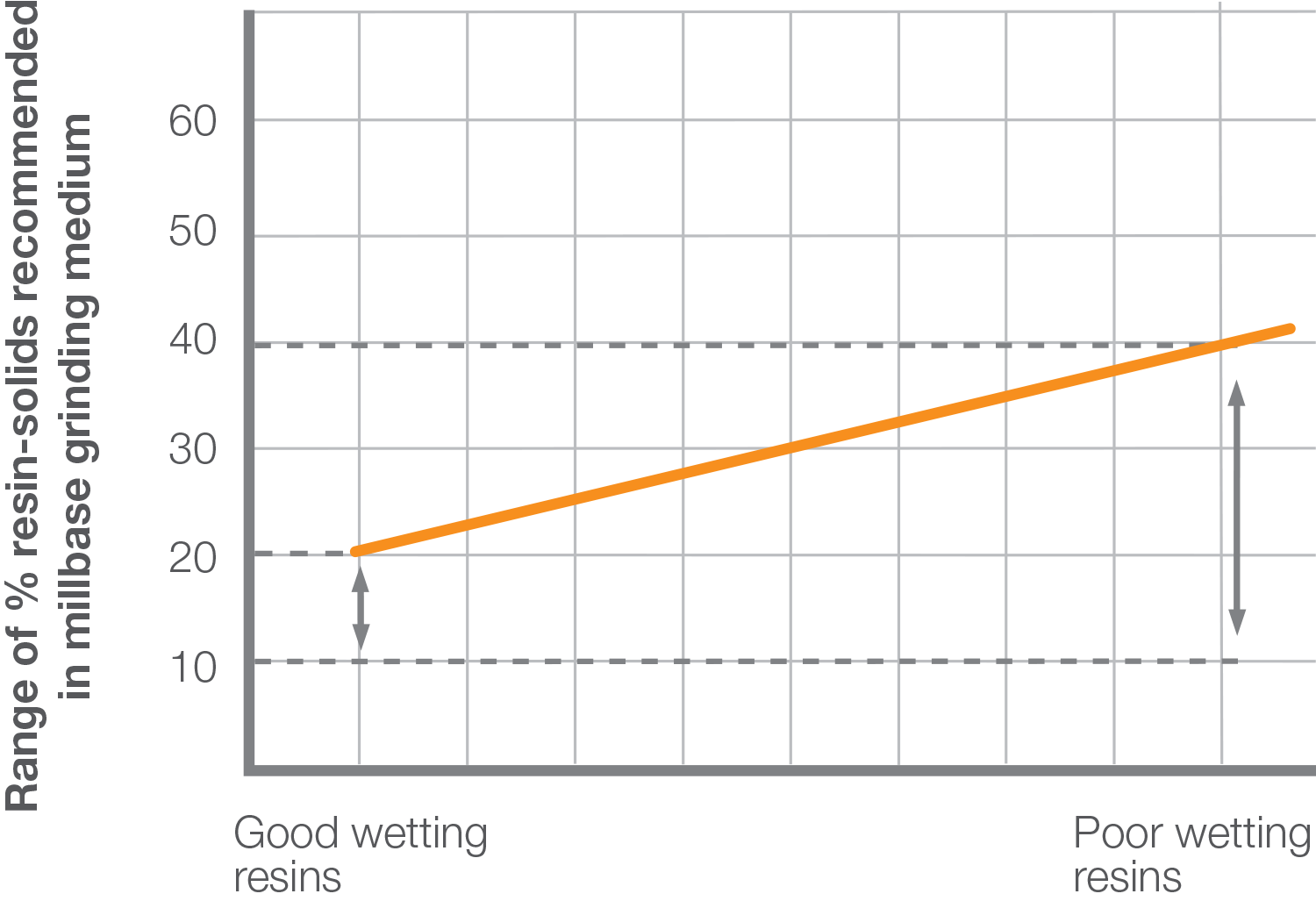
Anchoring of the dispersant to the pigment surface can be affected by competition between the resin and the dispersant for the surface of the particle. Once the dispersant anchor group is attached to the pigment surface, it will remain firmly attached. Molecules of resin, however, are transiently adsorbed on the surface of the pigment, and even though not firmly anchored they can still hinder the dispersant anchoring process. The competition for the pigment surface, resulting from using “good wetting” resins, is the most severe case.
ELECTROSTATIC STABILIZATION
ELECTROSTATIC STABILIZATION
Classic colloidal science explains electrostatic stabilization in terms of an electrical double layer. A charge is generated on the pigment surface, and a more diffused cloud of oppositely charged ions develops around it. As two particles approach each other, the charge provides a barrier to closer particle interactions.
Electrostatic stabilization is effective in media of reasonably high dielectric constant, principally water; although even in water-based systems, steric stabilization, or a combination of steric and charge stabilization, will often provide better performance. Therefore, both mechanisms of stabilization are found in the Solsperse™ Hyperdispersants for use in aqueous systems.



STERIC STABILIZATION
Charge stabilization will not be effective in media of low dielectric constant (the vast majority of organic solvents and plasticizers), and steric stabilization is required to maintain dispersed particles in a stable non-flocculated state. Steric stabilization relies on the adsorption of a layer of resin or polymer chains on the surface of the pigment. As pigment particles approach each other, these adsorbed polymeric chains intermingle and lose a degree of freedom. In thermodynamic terms, this is expressed as a reduction in entropy.
Alternatively, it can be considered that, as the chains intermingle, solvent is forced out from between particles. This leads to an imbalance in solvent concentration that is resisted by osmotic pressure which forces solvent back between the particles.


Stage 1
Stage 2
Stage 3
Stage 4
08
Formulating a Solsperse™ Hyperdispersant Millbase
CALCULATION OF %AOWP OF POLYMERIC DISPERSANT
The theoretical amount of polymeric Solsperse™ agent required in a millbase is 2 mg of polymeric agent per square meter of pigment surface area.
Example: Pigment surface area – 70 m /g. Therefore, 140 mg polymeric agent per 1 g pigment are required = 14 g agent/100 g pigment, i.e. 14% AOWP. Synergists (if required) are used with the polymeric agent in ratios between 4:1 and 9:1 polymeric:synergist.
DETERMINES THE OPTIMUM AMOUNT OF SOLSPERSE HYPERDISPERSANT
(Can be performed on lab. shaker e.g. Red Devil) Using the higher pigment content established in Stage 2, carry out a series of polymeric agent dosages around the theoretical %AOWP (+ any synergist required) to optimize the dosage.
Determine best dispersant dosage by measuring desired property.
OPTIMIZES THE FINAL PIGMENT CONCENTRATION
(Should be done in equipment representative of bulk production) Using the established in Stage 3, prepare final ladder series of pigment contents - maintaining agent:pigment ratio determined in Stage 3 to determine optimum amount which gives best final product.
©2025 The Lubrizol Corporation. All Rights Reserved.
Learn More About Lubrizol Dispersants


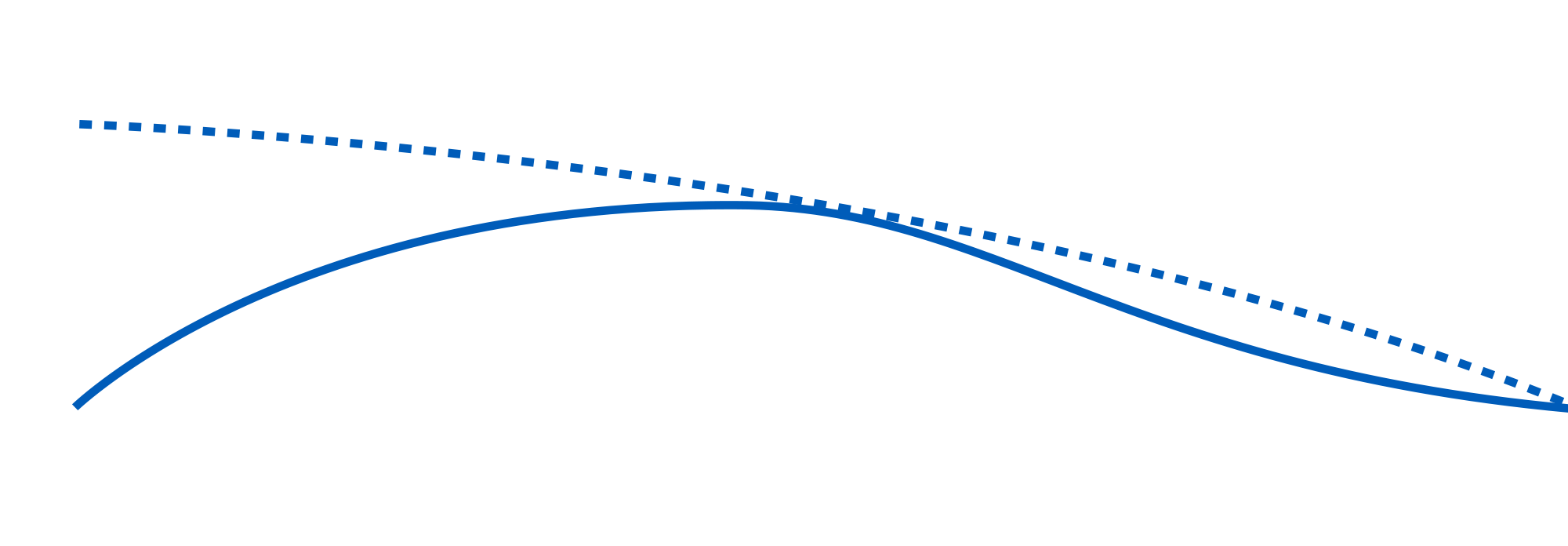
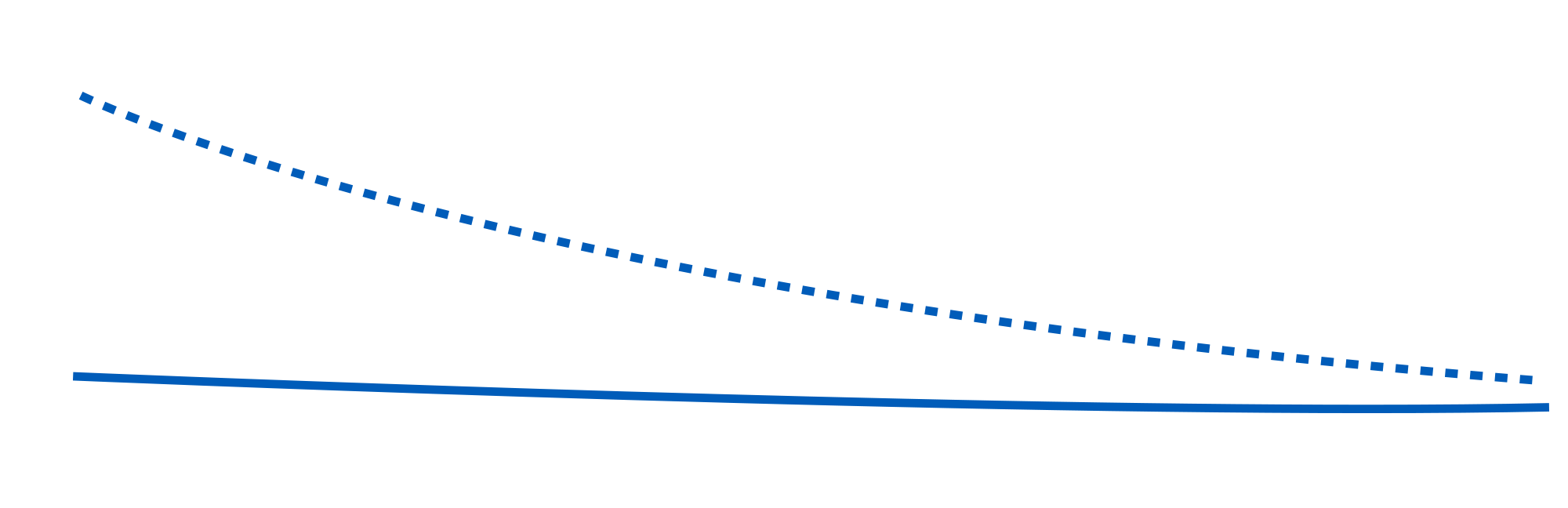
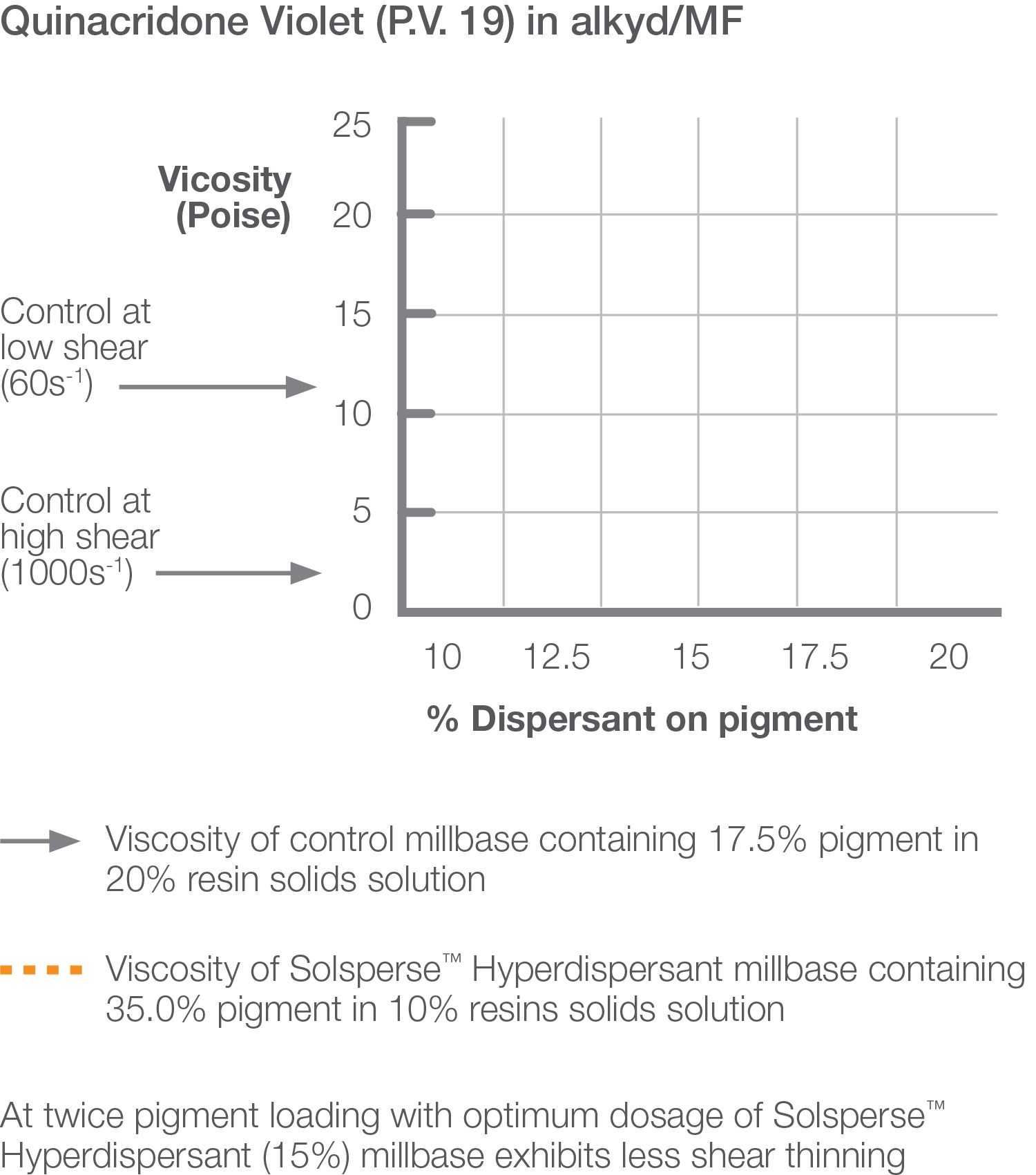
One fundamental requirement of steric stabilization is that the chains are fully solvated by the medium, meaning the chains will be free to extend into the medium. In systems where the chains are not so well solvated they will prefer to lie next to each other on the surface of the pigment, providing a smaller barrier to interparticle attraction. The greater steric repulsion generated by Solsperse™ Hyperdispersants creates the minimum movement in the potential energy curve (based on the DLVO theory), thus reducing overall viscosity.


POLYMERIC CHAINS
The polymeric chains are optimized to cover a wide range of Hansen solubilities, using several different polymer chain types and ensuring compatibility across many different functional binder systems. This need for compatibility extends throughout the final drying stages of any applied coating and in the dry film. If it ceases, flocculation may occur, leading to losses in gloss and tinctorial strength or haziness in the film.
The molecular weights of the Solsperse™ Hyperdispersants products provide polymer chains of optimum length to overcome Van der Waals’ forces of attraction between pigment particles. If the chains are too short, then they will not provide a sufficiently thick barrier to prevent flocculation.


There is generally an optimum chain length over and above which the effectiveness of the stabilizing material ceases to increase; indeed, in some cases, molecules with longer than optimum chains can be less effective. Ideally the chains should be free to move in the dispersing medium, so chains with anchor groups at only one end have shown to be the most effective.
Single anchor, single chain structures are polymers with terminal function groups. They are fast wetting and good with grind resins and, by their nature, only contain one type of anchoring group; therefore, they are most effective on a single class of pigments. Multi-anchor or multi-chain structures, also known as comb copolymers, are better for long-term stability and broad pigment compatibility as they can contain multiple types of anchor technology.


Chroma
Milling Time (mins)
180
10
60
Chroma Development
50
48
46
44
42
40
38
36
34
Control
Solsperse

Good “wetting” or “grinding” resins are not essential for use in the dispersion medium in the presence of Solsperse™ Hyperdispersants since they themselves will provide the essential pigment wetting functions. Other resins, more suitable to end-film properties, can be used.

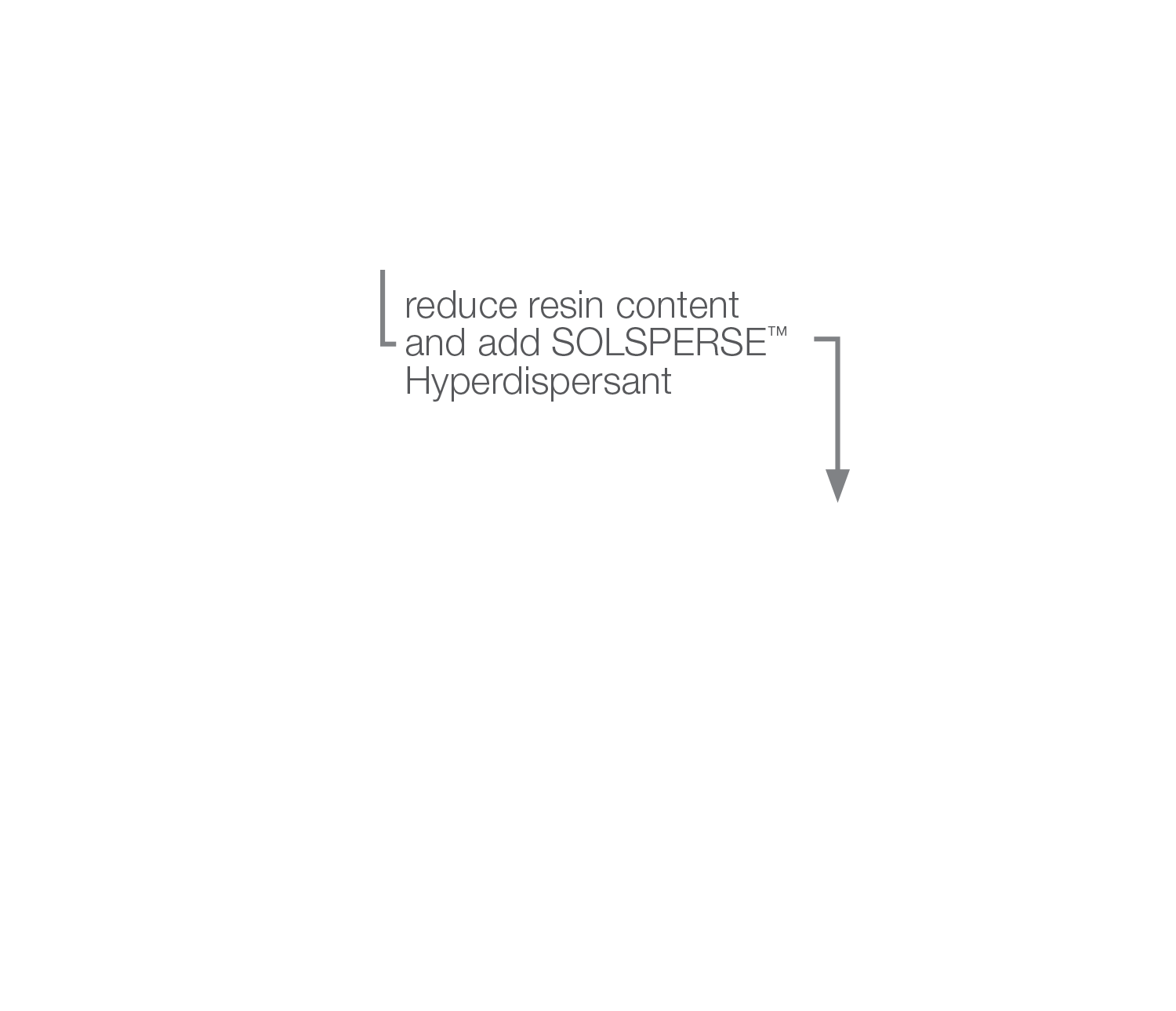
We recommend that minimum resin solids are used in the millbase, but in sufficient quantities to produce the necessary let-down stability, or to produce final coatings with the appropriate resin and solvent contents. If relatively good wetting resins are used, we recommend using a 10% to 20% resin solids solution as the grinding medium, and for relatively poor wetting resins, the minimum amount up to the level conventionally used.
DETERMINES THE HIGHER PIGMENT CONTENT REQUIRED
(Can be performed on lab. shaker e.g. Red Devil) Using the %AOWP of polymeric agent (from Stage 1) + synergist (if required), prepare a series of millbases with increasing pigment contents in a grinding medium containing approximately 10% solid resin.
Note: the ratio of dispersant to pigment must be maintained. The pigment concentration giving the same viscosity as the control should now be used in Stage 3.
SURFACE ACTIVE AGENTS
As with resins, surface active agents, such as surfactants and anti-foams, may also compete for the surface of the pigment so may have to be omitted from the millbase or added during let-down.
Similarly, matting agents and anti-settling agents should be added at the let-down stage because dispersants may have adverse effects on them if incorporated in the millbase.
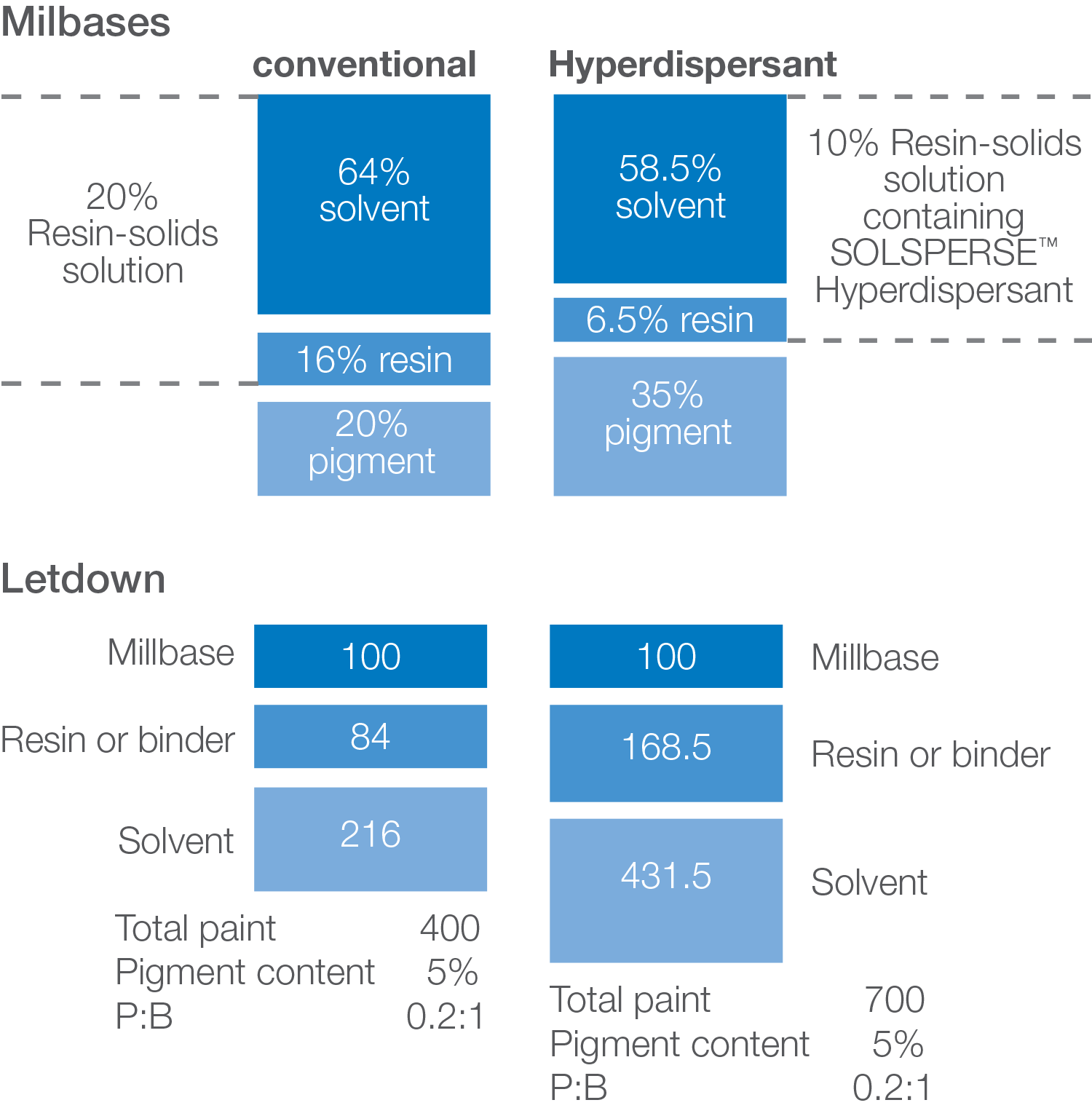
HIGHER PIGMENT CONCENTRATION IN THE MILLBASE
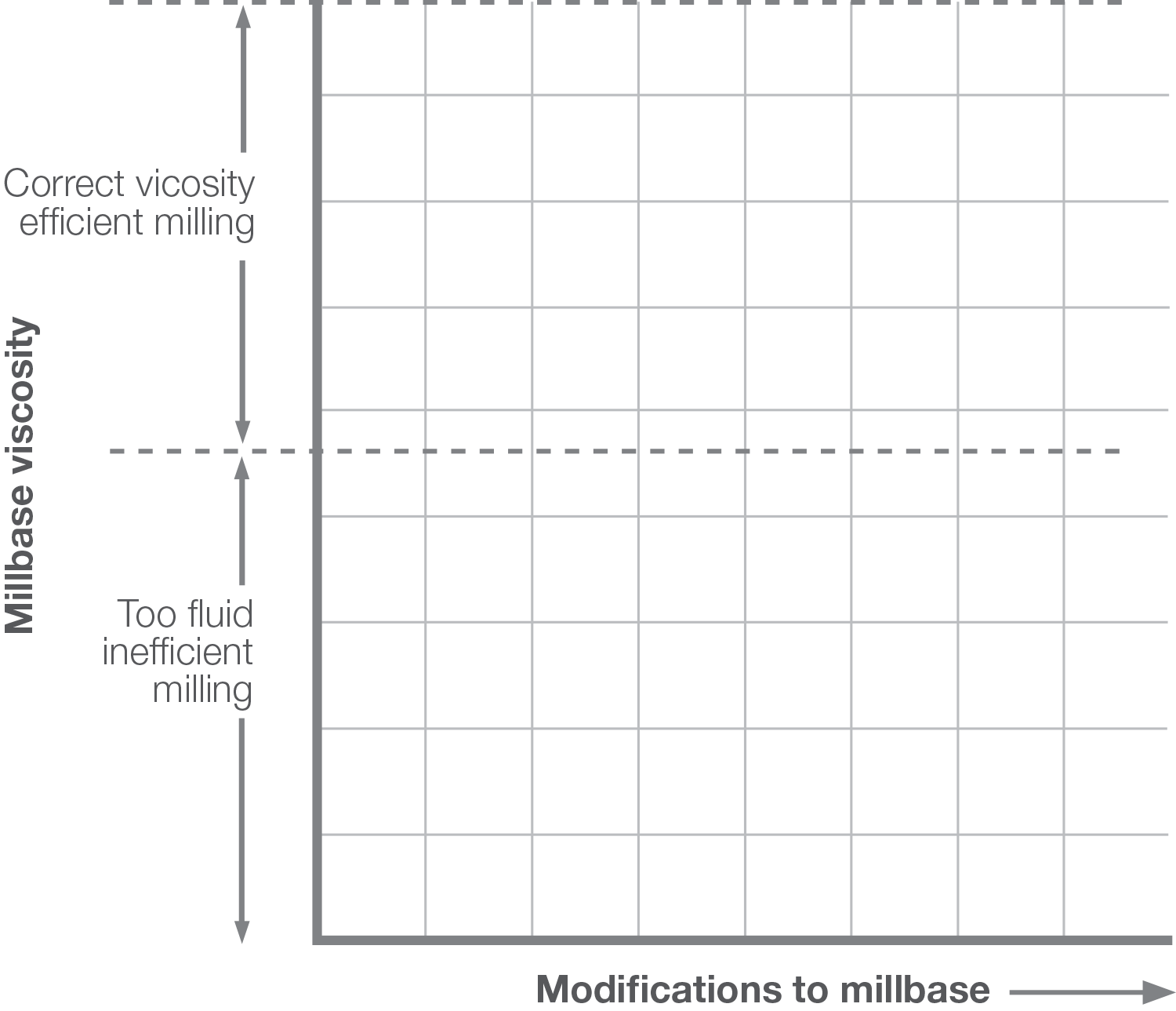
In minimizing the amount of resin in the millbase when using Solsperse™ Hyperdispersants contribution from the resin to the viscosity of the millbase is reduced as is the viscosity contribution from the pigment.
In low resin or resin-free systems the viscosity may be too low to generate sufficient shear forces for effective dispersion. In these cases, the pigment content should be increased.
BETTER MILLBASE RHEOLOGY
Millbases containing Solsperse™ Hyperdispersants exhibit lower viscosity, which allows higher pigment loading whilst maintaining a workable dispersion viscosity, often combined with improved flow.
REDUCED VOLATILE ORGANIC CONTENT (VOC) IN HIGH-SOLIDS COATINGS
The rheology of a coating is made up of contributions from the solvent, resin and pigment. In “normal” low-solids coatings the contribution from the pigment is dominated by that of the resin solution.
However, in high-solids coatings less solvent is present and the resins used are of low viscosity, therefore the contribution from the pigment is far more significant. Solsperse™ Hyperdispersants, by reducing interparticulate attractive forces, dramatically lower the pigmentary contribution towards viscosity.
Therefore, when comparing two high-solids coatings, one with, and one without dispersant, the former will have lower viscosity, or more usually, less solvent at equal application viscosity.
GREATER PRODUCTIVITY
Being able to disperse more pigment in a given time and thereby produce more final product from a given weight of millbase leads to productivity gains.
GREATER MILLBASE FLEXIBILITY
Solsperse™ Hyperdispersants permit higher pigment loadings in the millbase, not only improving productivity, but also reducing the amount of potentially incompatible medium that is carried through to the final coating formulation.
Solsperse™ Hyperdispersant millbases, therefore, have wider acceptance in a range of base finishes, especially if a wide compatibility resin can be used; important for the production of tinters for mixing schemes.
In addition, since dispersants act as wetting agents, there is less need to select grinding media purely for their wetting power: the formulator can be more flexible in the choice of dispersing medium.
HIGHER COLOR STRENGTH
A reduction in the average size of pigment particles leads to increases in color strength. A higher pigment content in the millbase increases the number of particle collisions, giving faster reduction of particle size. Dispersants permit milling at higher pigment concentrations, causing the more rapid breakdown of particles; at the same time, they will prevent viscosity building up during the grinding process.
Finally, they will impart colloidal stability to the finer particles so that they do not flocculate and their full intrinsic color strength is utilized.
HIGHER GLOSS
A maximum particle size greater than five microns is generally considered unacceptable for gloss paint finishes, whilst in high quality gloss coatings, three microns is the acceptable limit. Most inks contain particles dispersed to less than one micron. Large particles, present in the coating, may occur either through a failure to disperse effectively or through flocculation or “shock seeding” during let-down.
Solsperse™ Hyperdispersants aid the efficiency of the grinding process, directly through their ability to wet out newly created pigment surface, or indirectly by increasing the millbase pigment concentration. Dispersants also reduce the incidence of large particles due to flocculation.
INCREASED BRIGHTNESS
The particle size distribution in a dispersed pigment system influences the brightness of a surface coating; the presence of large particles tends to introduce a “dulling” effect.
Solsperse™ Hyperdispersants, because of their ability to produce a narrower particle size distribution, give brighter colors.
BETTER FLOCCULATION RESISTANCE
Flooding and flotation of surface coatings are dependent on the relative densities, sizes and state of dispersion of pigment particles in a system therefore they are adversely affected by pigment flocculation. Although dispersants cannot affect the relative densities of pigments, they are able, through effective stabilization, to prevent flocculation. This improvement in flocculation resistance not only leads to better optical properties, but also to improved flooding and flotation properties; and thereby a general improvement in the reproducibility of a coating by different application methods.








Polymer Design
Polymer Chain Length for Optimal Steric Stabilization
Control Millbase
Solsperse™ Hyperdispersant Millbases






40%
35%
30%
25%
20%

20%
18%
16%
14%
12%
Theoretical % AOWP


34%
32%
30%
Polymeric Chain
Creates steric barrier
Anchoring Segment
Adsorbs onto pigment
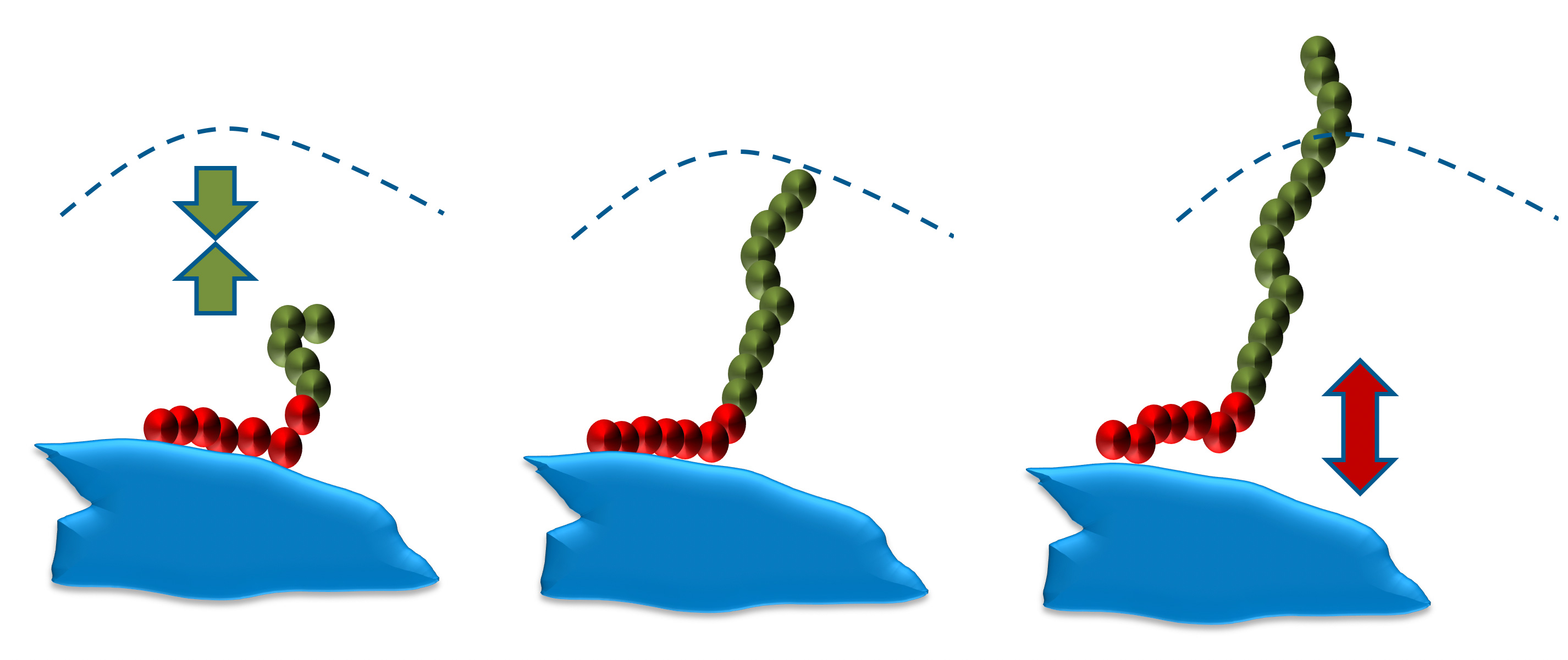
Too short = no steric barrier

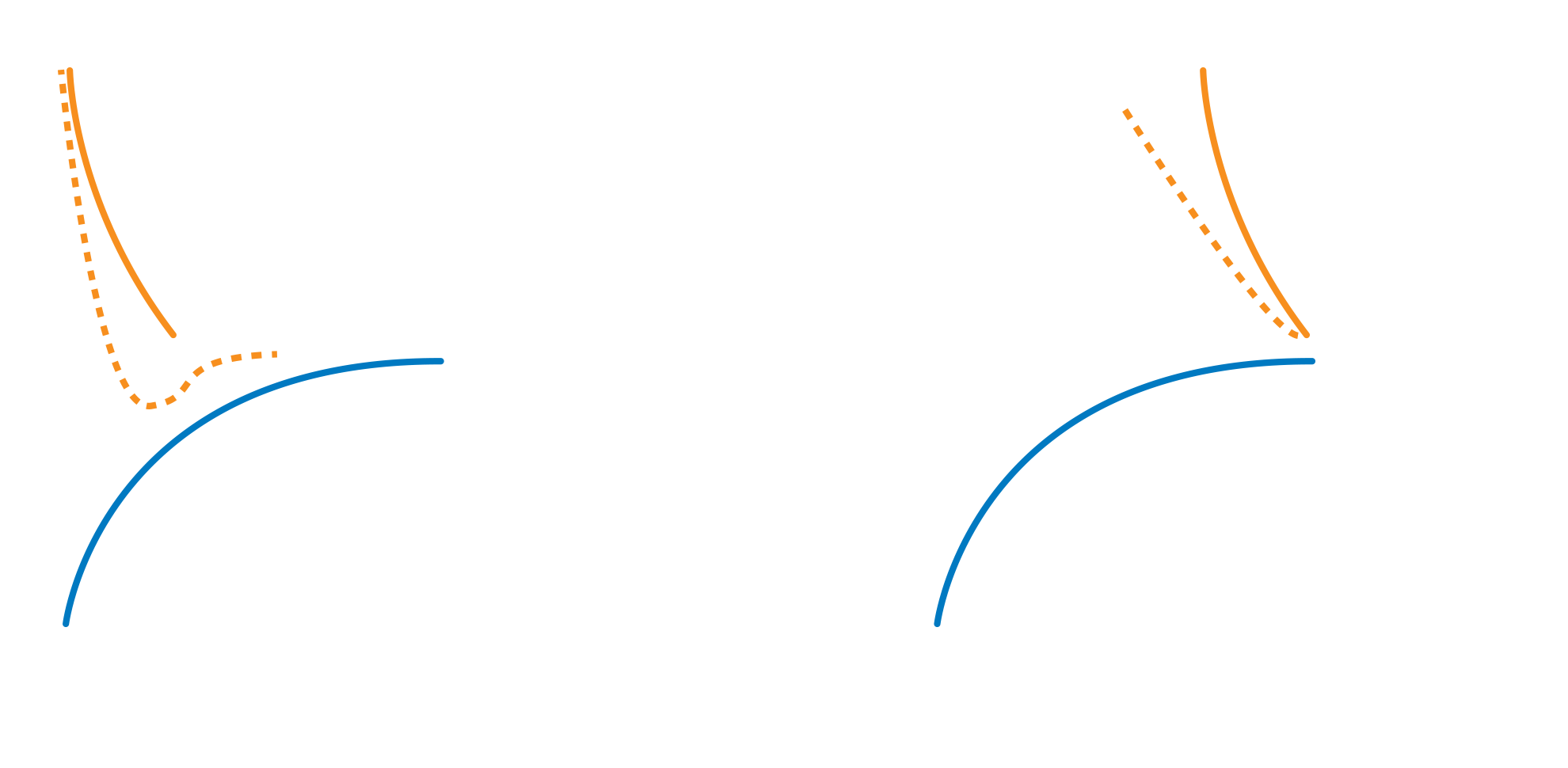

Effect of Solplus™ L400
32.5% CNTRL
50% CNTRL
50% Sol L400
For digital applications the pigment surface area can often be considerably higher and therefore the dosage may need to be increased. If required, the amount of Solsperse™ synergist should be additional to the amount of polymeric Solsperse™ Hyperdispersant.
Dispersant
Grind Resin
?
Dispersant
Synergist


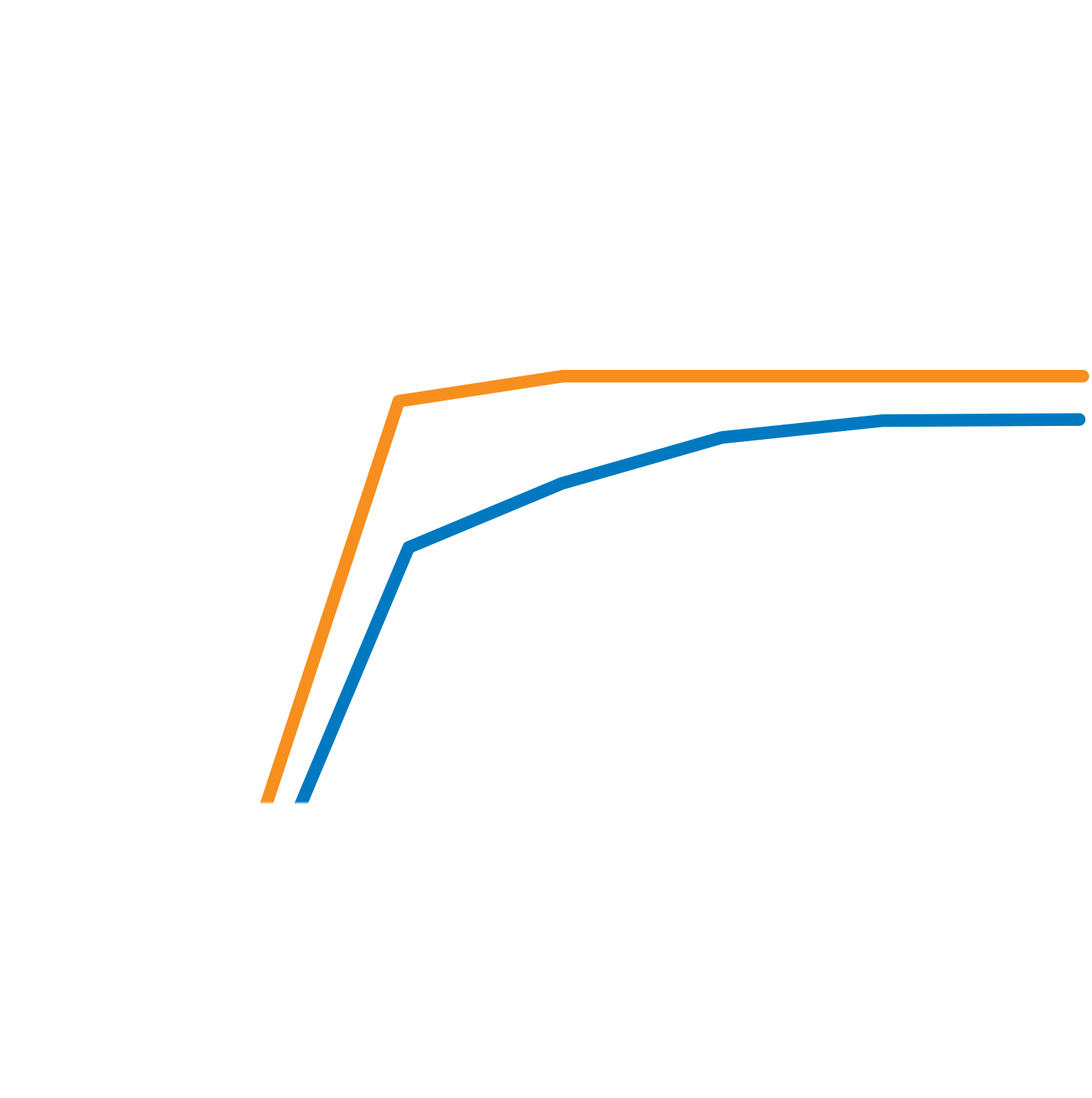




2/12
1/12
3/12
4/12
5/12
6/12
7/12
8/12
9/12
10/12
11/12
12/12
Follow Lubrizol Performance Coatings On LinkedIn
Visit Us On YouTube

Learn More

Learn More
Learn More

CONTENTS
01 Why are Dispersants Important?
02 The Role of Hyperdispersants
03 Background to Effective Stabilization
04 The Design of Dispersants
05 Anchor Groups
06 Practical Demonstration of Need for Correct Dosage
07 Formulation Guidelines When Using Solsperse™ Hyperdispersants
08 Formulating a Solsperse™ Hyperdispersant Millbase


Back to the Top

Back to the Top

Back to the Top

Back to the Top

2
Scroll
